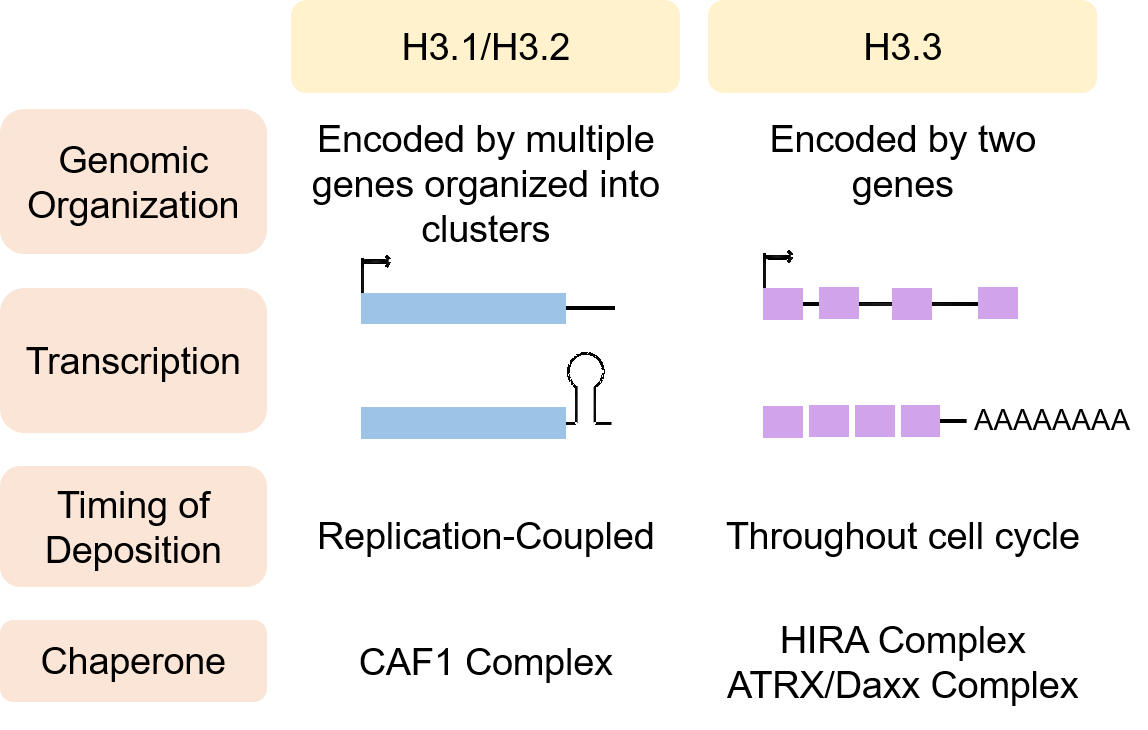
Chromatin remodeling is one of the first line of cellular reprograming that enables complex cell fate decisions. As such, epigenetic changes are essential to both the aging and the tumorigenic processes. Histone variants and histone chaperones have become the latest addition to a growing list of epigenetic regulators. Histone exchange allows chromatin to be highly responsive to changes in the environment, which are particularly dynamic throughout the tumorigenic process. Among the histone variants known thus far, histone H3.3 has been gaining interest in the chromatin field owing to its unique features. Unlike its canonical counterparts H3.1/H3.2, H3.3 incorporation into chromatin is cell-cycle independent, and it can be deposited at replication sites when canonical H3.1/H3.2 deposition is impaired. H3.3 has also been consistently associated with an active state of chromatin. Importantly, old age promotes the decline of canonical histones and the accumulation of histone H3.3 suggesting that H3.3 may play a pathological role in age-related diseases.
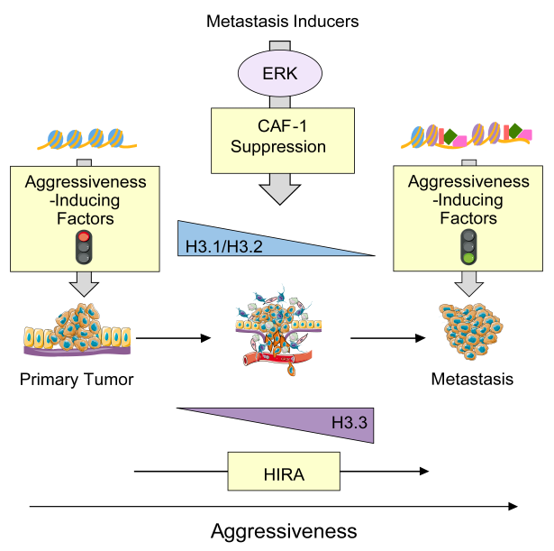 Our previous work has shown that metastasis-inducing pathways regulate histone chaperones to reduce canonical histone incorporation into chromatin, triggering deposition of H3.3 variant at the promoters of poor-prognosis genes and metastasis-inducing transcription factors. This specific incorporation of H3.3 into chromatin is both necessary and sufficient for the induction of aggressive traits that allow for metastasis formation and underscores the importance of H3 variants for cell fate decisions in cancer. The Gomes lab is working on expanding these studies to evaluate the contribution of Histone H3 variants to other aspects of the tumorigenic process.
Our previous work has shown that metastasis-inducing pathways regulate histone chaperones to reduce canonical histone incorporation into chromatin, triggering deposition of H3.3 variant at the promoters of poor-prognosis genes and metastasis-inducing transcription factors. This specific incorporation of H3.3 into chromatin is both necessary and sufficient for the induction of aggressive traits that allow for metastasis formation and underscores the importance of H3 variants for cell fate decisions in cancer. The Gomes lab is working on expanding these studies to evaluate the contribution of Histone H3 variants to other aspects of the tumorigenic process.
Questions of Interest: